Pharmazeutische Wassersysteme
Wasser ist ein wesentlicher Bestandteil jeder pharmazeutischen Verarbeitung. Die Sicherstellung einer konstanten Versorgung mit qualitativ hochwertigem Wasser, das mit der erforderlichen Durchflussmenge und Temperatur an den Verwendungsort geliefert wird, ist eine grundlegende Anforderung. Alfa Laval Geräte für pharmazeutische Wassersysteme sorgen für einen problemlosen Betrieb.
Erfüllung der Anforderungen an pharmazeutische Wassersysteme mit innovativen Lösungen
- Optimieren Sie die Bakterienkontrolle
- Problemlose Installation und Validierung
- Aufrauhung verhindern
- Minimierung von Wartung und Desinfektion
Wenn Sie ein Pharma- oder Biotech- Hersteller sind, der Rouging, Biofilmbildung oder andere Verunreinigungen in seinen pharmazeutischen Wassersystemen verhindern möchte, kann Alfa Laval Ihnen helfen, die strengen Hygieneanforderungen zu erfüllen.
Mit Alfa Laval ist es einfach, alle pharmazeutischen Anforderungen zu erfüllen. Profitieren Sie von unserem umfassenden Fachwissen über pharmazeutische Prozesse, unserer umfangreichen und bewährten Hygieneausrüstung und unserem weltweiten Servicenetz.
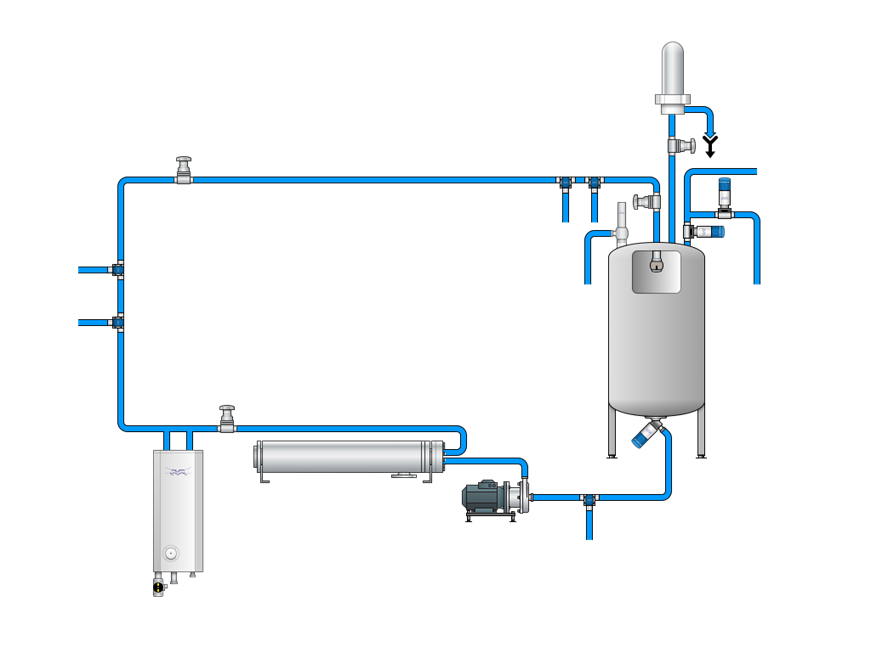
System-Übersicht
Wasser ist in der pharmazeutischen Produktion lebenswichtig. Nach der Aufbereitung muss es über validierte, hygienische Systeme gelagert und verteilt werden. Sehen Sie sich das Prozessdiagramm an und erfahren Sie, wie die Lösungen von Alfa Laval jede Phase der pharmazeutischen Wassersysteme, einschließlich der WFI-Wassersysteme, unterstützen. Sie können die Tabelle auch als PDF herunterladen.
Dokumente
Einfache Validierung und Wartung: Alfa Laval Q-doc
Für eine problemlose Qualifizierung und Validierung gibt es Alfa Laval Q-doc, unser hochmodernes Dokumentationspaket für die Pharmaindustrie. Q-doc umfasst jeden Aspekt der Produktion, von der Beschaffung des Rohmaterials bis zur Auslieferung der fertigen Ausrüstung. Dies sorgt für volle Transparenz und ermöglicht die Rückverfolgung der kleinsten Material- oder Herstellungsänderung - selbst bei Ersatzteilen.

Kontaktformular
Wir möchten Sie bestmöglich unterstützen. Füllen Sie deshalb bitte alle Pflichtangaben aus, damit wir Ihnen schnellstmöglich auf direktem Weg antworten können.
Wichtige Information
Private Anfragen werden von uns nicht bearbeitet. Unsere Vertriebspartner helfen Ihnen gern, einen Installateur in Ihrer Nähe zu finden.

